Composition Analysis
Organic and Polymer Materials
Evaluation of Organic and Polymer Materials
Organic materials are a highly complex substance comprising from a combination of rather limited number of elements of carbon, hydrogen, nitrogen, oxygen, and a few others and possessing a huge variety of properties.
In the analysis of organic materials JET-TEC can provide not only the structural analysis of these materials themselves but also the analysis of organic substance derived from the materials such as elution components and emission gases from raw materials and products.
JFE-TEC has expertise and experience with selection of analytical procedures and the data analysis in this field and is always willing to consult with the clients before starting any evaluation of the organic materials to solve the problems.

Instruments and Analytical Examples
JFE-TEC uses the following instruments for the analysis of organic and polymer materials.
| Classification | Instrument name | Abbreviation | Outline of analysis | |
|---|---|---|---|---|
| Qualitative and quantitative analysis | Liquid chromatography system | Photodiode array detector | HPLC-PDA | The above analytical method is used for the quantitative analysis of organic components. The UV-Vis detector, the fluorescence detector, and the differential refractive index detector are used for detection and quantification of the UV and visible light absorbing components, for detection and quantification of the fluorescent components, and for detection and quantification of the components by change of diffractive index, respectively. These detectors are selected depending on the properties and the concentration of quantified components. |
| UV-Vis detector | HPLC-UV/Vis | |||
| Fluorescence detector | HPLC-FL | |||
| Differential refractive index detector | HPLC-RI | |||
| andem mass spectrometer | LC-MSMS | |||
| Gel permeation chromatography system | GPC | This method is used for measurement of the molecular weight distribution of polymer materials. | ||
| Gas chromatography system | Thermal conductivity detector | GC-TCD | This analytical method is used for the quantitative analysis of organic components. These detectors are selected depending on the components to be analyzed, and the thermal conductivity detector, the flame ionization detector, the flame photometric detector, and the electron capture detector are used for the identification and the quantification of inorganic gas components such as nitrogen and oxygen, for the identification and the quantification of hydrocarbons, for the identification and the quantification of phosphorous and sulfur in organic compounds, and for the identification and the quantification of alkyl mercury and PCB, respectively. | |
| Flame ionization detector | GC-FID | |||
| Flame photometric detector | GC-FPD | |||
| Electron capture detector | GC-ECD | |||
| Gas chromatography mass spectrometer | GC-MS | The gas chromatography mass spectrometer is the most versatile instrument for qualitative and quantitative analysis of organic components. The gas chromatography mass spectrometry can be coupled with the sample introduction system such as the headspace (HS) sampler, the purge and trap (P & T) system, the automatic thermal desorption (ATD) system, and the pyrolysis (Py) system to broaden its analysis capability. | ||
| Elemental analysis instrument | C, H, N analysis instrument | - | These elemental analysis instruments are used for the elemental analysis of coals and resins | |
| O or S analysis instrument | - | |||
| Structural analysis | Fourier transform infrared spectroscopy instrument | FT-IR | The Fourier transform infrared spectroscopy is suitable for analysis of foreign particlesbecause the specific wavelength and absorption intensity found in the infrared spectra may suggest the particular functional group in the sample. Use of an infrared microscopy unit permits the analysis of trace foreign particle. | |
| Raman laser and spectrometer | - | A sample is irradiated with a laser source at particular wavelength to scatter a small fraction of the incident radiation (Raman scattering light) and to shift at the frequencies that correspond to the vibrational transition specific to the sample, providing the information on the molecular structure. This method can be applied for identification of foreign particles in the sample and is very powerful for non-destructive failure analysis of resin films and SiC films. | ||
| Fourier transform nuclear magnetic resonance spectrometer | FT-NMR | A sample is immersed in a strong magnetic field and hit with radio waves to generate the interaction with the nuclei spin and nearby electron causing the signal to absorb at a slightly different frequency than for a single atom. This difference is used to get the information on the chemical bonding of molecules so that FT-NMR is useful for the structural analysis of organic and polymer materials. JFE-TEC has probes for solid-state NMR spectroscopy and can perform the solid-state NMR spectroscopy. | ||
| Thermal analysis | Thermogravimetric differential thermal analyzer | TG-DTA | The thermogravimetry (TG) is a measurement of a weight change when heating a sample, the differential thermal analysis (DTA) is to measure any temperature difference between a sample and an inert reference under identical thermal cycle and to record heat changes in the sample, exothermic or endothermic, and the differential scanning calorimetry (DSC) is to measure heat effects associated with phase changes of the sample and chemical reactions as a function of temperature. These thermal analyses are very informative methods in physical characterization of the thermal behavior of organic and polymer materials. | |
| Thermogravimetric mass spectrometry instrument | TG-MS | |||
| Differential scanning calorimetry instrument | DSC | |||
Example of Fourier Transform Infrared Spectroscopy Instrument (FT-IR)
Upon irradiation of a sample with infrared light the amount of light absorbed (or transmitted) by the sample is measured as a function of its wavelength. The specific wavelength and the intensity of the infrared spectrum provides the information on the type of chemical bonds of the sample.
Use of FT-IR also gives useful information to find whether the sample is the foreign particles or not.
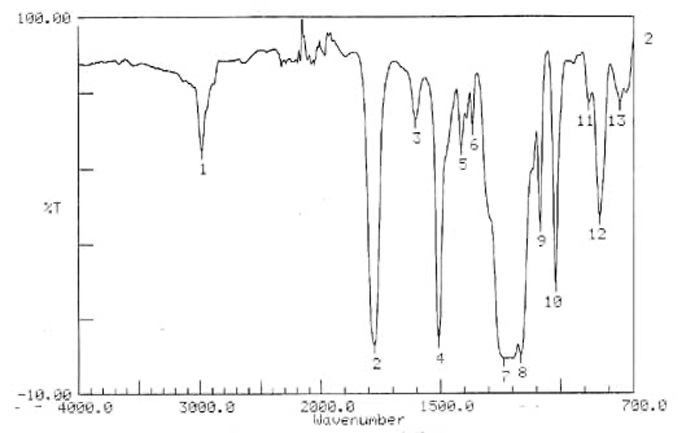
Analysis of Foreign Particles
The figure below demonstrates an FT-IR spectrum of resin particles as contaminants. .
The absorption peaks and their intensity of the resin particles in the FT-IR spectrum below match very well to the FT-IR spectrum of polycarbonate, clearly indicating that the foreign particle is polycarbonate.
Nuclear Magnetic Resonance (NMR)
A sample is immersed in a strong magnetic field and hit with radio waves to generate the interaction with the nuclei spin and nearby electron causing the signal to absorb at a slightly different frequency than for a single atom. This difference provides the information on the chemical bonding of molecules so that FT-NMR spectroscopy is very useful for the structural analysis of organic and polymer materials. Typical nuclei analyzed by FT-NMR are hydrogen and carbon. As compared to the analysis by FT-IR spectroscopy FT-NMR spectroscopy generates more detail information on the chemical structure, in particular the relative number of hydrogen atoms can be determined allowing further detail characterization of the chemical structure. Application of FT-NMR spectroscopy is further expanded to a broader field including a two-dimensional NMR method and the distortionless enhancement by polarization transfer (DEPT) technique.
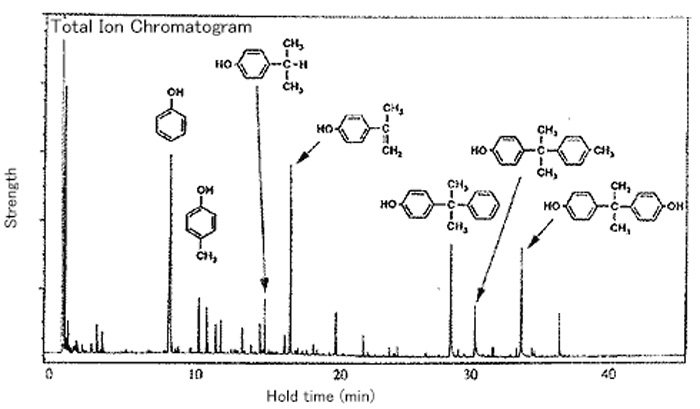
Example of Pyrolysis Gas Chromatography Mass Spectrometry (Py-GC-MS)
Pyrolysis-gas chromatograph-mass spectrometry (Py-GC-MS) uses a combination of the instantaneous thermal decomposition method with the evolved gas analysis and makes the characterization of the intractable and involatile materials easier, while the conventional gas chromatography cannot analyze these materials.
This method also allows characterization of organic materials insoluble in a solvent.
Structural Analysis of Epoxy Resin
The figure below demonstrates the Py-GC-MS spectrum of a bisphenol A type epoxy resin as the example of analysis of resins insoluble in solvents.
While the analysis of this resin with the infrared spectroscopy can identify only a certain functional groups, the Py-GC-MS clearly identifies the chemical structure of various bisphenol A components in the epoxy resin.